【Article Information】
Finely control the intermolecular interactions and molecular stacking of non-condensed ring electron acceptors through halogen transposition strategy to achieve high-performance organic solar cells
First author: Gu Xiaobin, Zeng Rui
Corresponding author: Huang Hui*, Zhang Xin*, Liu Feng*
Unit: University of Chinese Academy of Sciences, Shanghai Jiao Tong University
【Research Background】
The invention of thick ring electronic receptors has greatly promoted the development of organic solar cells. At present, the photoelectric conversion efficiency of single -knotting devices has exceeded 20%. However, the high-ring electron receptor has a highly thick π-conjugate skeleton, which leads to the long-term synthetic routes and high production costs, which limits the commercial application of such materials. In contrast, non -thick ring electron receptors have attracted much attention for its simple molecular structure and simple synthetic route. The introduction of non -covalent "conformity locks" or large -scale blocking side chains enables the non -thick rings to achieve high levels and rigidity comparable to the thick ring structure. Applications for organic solar cells are possible. However, it is currently lacking the method of building an orderly aggregation state structure of non -thick ring electron receptors. Therefore, exploring the strategy of effective regulation of the aggregate state structure is essential for improving the efficiency of photovoltaic devices based on such materials.
【Introduction】
Recently, the research group and Professor Liu Ye of Shanghai Jiaotong University collaborated on an internationally renowned journal ANGEW. Chem. Int. Ed. Tors via Halogen Transposition for High-Performance Organic SOLAR CLLS's research paper. This work finely regulates the interaction between the molecular receptor of the non -thick ring electron receptor through the halogen conversion strategy, and realizes the transformation of two -dimensional layers to three -dimensional interoperability network aggregation structure. The binary and ternary organic photoresmal devices based on 3TTB-CLF obtained 17.46%and 18.24%of the photoelectric conversion efficiency of 17.46%, which refreshed the efficiency records currently based on non-thick ring electronic receptor organic photoretical components.
【Key points of this article】
Key point 1: Halogen transposition strategy
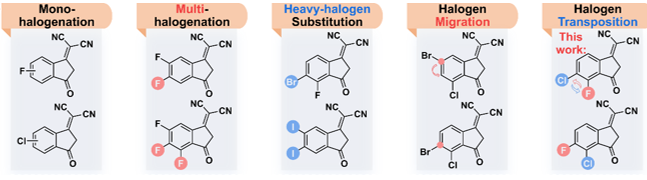
Figure 1. The literature has been reported and the end -based halogenization strategy proposed in this work
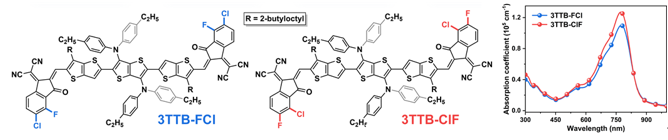
Figure 2. Chemical structure and film absorption spectrum
This work proposes the end-based conversion strategy, that is, the replacement position of the halogen atom is interchangeable, and the two new non-thick ring electronic receptor 3TTB-FCL and 3TTB-CLF (Figure 1-2) are designed and synthesized. UV-visible absorption spectrum display 3TTB-CLF's light absorption coefficient (1.27 × 105 cm−1) in solid state is significantly higher than 3TTB-FCL (1.10 × 105 cm−1), indicating that there is a stronger interaction between its molecules and more Tightly stacked.
Key Point 2: Regulating Intermolecular Interactions and Stacking
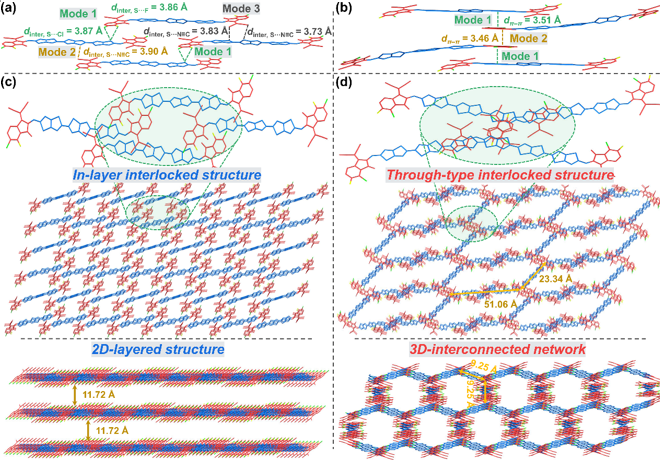
Figure 3. Research on the interaction and accumulation behavior based on crystal -based molecules
The results of the single crystal XRD diffraction analysis show that the 3TTB-FCL has a layer-locking structure that mediated the non-co-price interaction of multiple stabiliated non-co-price interactions, thereby forming a two-dimensional layered clustering structure. Instead, 3TTB-CLF consists of a penetrating inter-locking structure by interaction between the inter-molecular π-π interaction, so the three-dimensional interoperability network aggregate structure is formed to build a high-speed channel for charge transmission.
Key points 3: Realize new records of photoelectric conversion efficiency
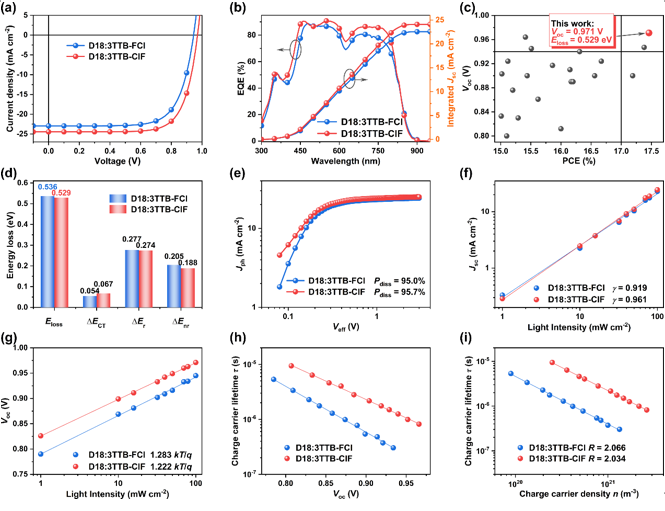
Figure 4. Photovoltaic performance and device physics research
Device physics and film morphology studies show that the D18:3TTB-ClF system has higher open circuit voltage, lower energy loss, more efficient exciton dissociation, more balanced charge transport and more suitable phase separation morphology, thus achieving a photoelectric conversion efficiency of 17.46%. It was further used as the third component to construct the D18:3TTB-4F:3TTB-ClF ternary device, achieving a record efficiency of 18.24% (Figure 3). This work provides an effective strategy for regulating intermolecular interactions and constructing efficient charge transfer channels. At the same time, the first breakthrough of 18% efficiency also shows the huge application potential of non-condensed ring electron acceptors.
Gu Xiaobin, a doctoral student in the research group, is the first author of this article, Zeng Rui, a doctoral student at Shanghai Jiao Tong University, is the co-first author, and Professor Huang Hui, Associate Professor Zhang Xin and Professor Liu Feng of Shanghai Jiao Tong University are the co-corresponding authors of the paper. The authors thank the National Natural Science Foundation of China, the Chinese Academy of Sciences and other related projects for funding.
【Link】
Precisely Regulating Intermolecular Interactions and Molecular Packing of Nonfused-Ring Electron Acceptors via Halogen Transposition for High-Performance Organic Solar Cells. Angew. Chem. Int. Ed. 2024, e202407355.
https://doi.org/10.1002/anie.202407355


